

เว็บพนันออนไลน์ gb69 ป๊อกเด้ง
gb69 เล่นพนันออนไลน์ที่มีคุณภาพและบริการที่ดีที่สุดสำหรับผู้เล่นทุกท่านทั่วโลกเพื่อความสนุกและความพึงพอใจของผู้เล่นทุกคนที่ชื่นชอบการเล่นพนันออนไลน์ในทุกๆ ครั้ง ที่นี่คือสถานที่เดียวที่คุณสามารถพบประสบการณ์การเล่นพนันออนไลน์ที่ยอดเยี่ยมและเป็นเอกลักษณ์ที่ไม่เหมือนใครทั้งหมด
ทำให้ gb69 สล็อต คุณจะได้สัมผัสประสบการณ์การเล่นพนันที่ยอดเยี่ยมจากเว็บไซต์อื่น ๆ ที่เคยเดิมพัน เลือกเดิมพันออนไลน์เสริมโอกาสให้ผู้เล่นสร้างรายได้และทำเงินอย่างง่ายดาย ด้วยคุณภาพเยี่ยมที่ไม่เหมือนใครในโลกของการพนันออนไลน์ที่ไม่มีวันหยุด เล่นพนันออนไลน์เสี่ยงโชค
ช่วยเพิ่มโอกาสชนะรางวัลเงินสดใหญ่และสร้างความสนุกและตื่นเต้นที่ไม่เหมือนใครได้ที่สามารถทำให้ผู้เล่นรู้สึกมั่นคงและสบายใจที่หลากหลาย สล็อต แตกหนัก แตกบ่อย เล่นเกมสล็อตออนไลน์ที่ทำให้มีโอกาสชนะรางวัลเงินสดและสร้างรายได้ในรูปแบบออนไลน์มากขึ้นสำหรับผู้เล่นทุกคน

เว็บตรงไม่ผ่านเอเย่นต์ เกมคาสิโนออนไลน์ที่ไว้วางใจ
โดยเฉพาะ gb69 slot เราเป็นเว็บไซต์การพนันที่ทันสมัยและมั่นคง ที่เพิ่มรายได้และปรับกฎเกณฑ์เพื่อเพิ่มความน่าสนใจในการเดิมพันออนไลน์อย่างแน่นอน ทำให้เราสามารถทำเงินได้อย่างมั่นคงและเป็นหนึ่งในเว็บไซต์ที่สร้างรายได้ เว็บไซต์การพนันของเราพร้อมต้อนรับผู้เล่นใหม่ทุกท่าน และเปิดรับผู้เล่นเข้ามาเล่นอย่างต่อเนื่องที่นี่ เว็บไซต์การพนันที่มอบประสบการณ์การเล่นที่สมจริงและไม่เบื่อด้วยระบบที่สะดวก และรองรับทุกระบบทำให้เล่นได้อย่างสะดวกสบาย
เพื่อให้ เว็บตรง ไม่ผ่านเอเย่นต์ gb69 autobet เว็บพนันออนไลน์ที่มุ่งมั่นที่จะให้ประสบการณ์การเดิมพันที่ดีที่สุดด้วยเทคโนโลยีทันสมัยและระบบมั่นคงที่ให้ประสบการณ์การพนันออนไลน์ที่ลื่นไหลและไร้ข้อจำกัดสำหรับผู้เล่นทุกคน ที่มีภาพกราฟิกคมชัดและเสียงเอฟเฟ็กต์ที่ดี ระบบเสถียรและเกมส์หลากหลายที่ไม่เบื่อได้สร้างรายได้ การเดิมพันออนไลน์ที่มั่นคงด้วยระบบที่ไม่มีข้อบกพร่องทั้งกราฟิกและเสียง เพลิดเพลินไปกับประสบการณ์การเล่นที่ดีที่สุด
มุ่งเน้นให้ผู้เล่นรู้สึกเหมือนอยู่ในคาสิโนแท้และทำให้ไม่รู้สึกเบื่อเล่นล่ะ รวมทั้ง สล็อต gb69 ค้นหาประสบการณ์การเล่นพนันออนไลน์ที่ปลอดภัยและสนุกสนานอยู่ที่นี่ จะท้าทายให้คุณต้องเล่นเองตอนนี้ ทุกรายละเอียดครบครันและพร้อมจะมอบความสนุกที่แท้จริงให้คุณ ไม่ต้องออกรถมาเล่นแล้ว

เล่นคาสิโนออนไลน์ที่มีความสนุกสุดพิเศษ
การเดิมพันออนไลน์ในประเทศไทยเป็นที่นิยมอย่างมากและมั่นใจได้ว่าคุณจะได้รับโบนัสอันใหญ่ทุกครั้ง เพื่อที่จะได้ gb69 ความสนุกสุดพิเศษของการเดิมพันออนไลน์ที่ทันสมัย สะดวกสบาย และสมบูรณ์แบบที่ไม่ต้องเข้าสู่ระบบซ้ำๆ บริการการพนันออนไลน์ที่ทันสมัยและมีโอกาสที่จะได้รับรางวัลมหาศาลทุกครั้ง ! เล่นพนันออนไลน์ที่ที่นี่เพื่อชนะรางวัลมากมายแบบทันที อีกทั้งยัง สล็อตแมชชีน สล็อตเว็บตรง gb69 สล็อต คุณสามารถเริ่มเดิมพันออนไลน์ทุกที่ทุกเวลาที่ต้องการด้วยความสะดวกสบาย
มั่นใจว่าคุณจะได้รับประสบการณ์ที่ดีที่สุดที่นี่ อีกทั้งยังสามารถเสี่ยงโชคออนไลน์เพื่อที่จะได้เงินเพิ่มเติมเนื่องจากความสะดวกสบายและเล่นได้ทุกที่ ตลอด 24 ชั่วโมง ควรศึกษาข้อมูลและเรียนรู้เทคนิคการเดิมพันออนไลน์เพื่อความปลอดภัยของคุณก่อนที่จะลงเดิมพันในเว็บไซต์การพนันออนไลน์ จึง gb69 เข้าสู่ ระบบ

เว็บไซต์การพนันออนไลน์ที่มีบริการอัตโนมัติ 24 ชั่วโมง
เว็บพนันทันสมัย ด้วยระบบฝากถอนอัตโนมัติ 24 ชั่วโมง ที่มั่นคง สนุกสนาน และหลากหลายลูกค้าชอบในปัจจุบัน. gb69 slot เพลิดเพลินกับการเล่นพนันออนไลน์ได้ตลอด 24 ชั่วโมงด้วยบริการอัตโนมัติที่สะดวกและไม่มีปัญหา เว็บพนันออนไลน์ที่มั่นคงและไม่ซับซ้อนในการจ่ายเงินรางวัลทุกรางวัลโดยตรงให้ผู้เล่นได้แน่นอน ไม่ต้องผ่านตัวแทน และระบบจ่ายเงินรางวัลให้ผู้เล่นได้แน่นอนในเกมของเรา เว็บไซต์ของเราทำให้คุณเล่นพนันออนไลน์ได้อย่างสะดวกและรวดเร็ว
ฝากถอนเงินได้ทันที 24 ชั่วโมง ไม่มีโอกาสผิดหวัง และ สล็อตทรูวอเลทเว็บตรง gb69 คาสิโนออนไลน์ ท่านมีโอกาสที่จะเพลิดเพลินกับประสบการณ์การเดิมพันออนไลน์ที่ท้าทายและสนุกสนานที่สุด ที่ไม่ควรพลาด! ท่านจะได้รับประสบการณ์การเดิมพันออนไลน์ที่ท้าทายและทำให้คุณตื่นเต้นมากขึ้นที่สุด
ในการเล่นเกมออนไลน์ที่ไม่เหมือนใคร!สามารถการันตีการจ่ายรางวัลให้กับนักพนันที่เข้ามาใช้บริการได้จริง กับระบบเกมส์ออนไลน์ของเรา ดังนั้นจึง เว็บพนันออนไลน์ของเรามั่นคง ด้วยระบบการเงินที่ปลอดภัย และมีโปรโมชั่นมากมายที่ทำให้คุณเพลิดเพลินและตื่นเต้นทุกครั้งที่เข้าเล่น!
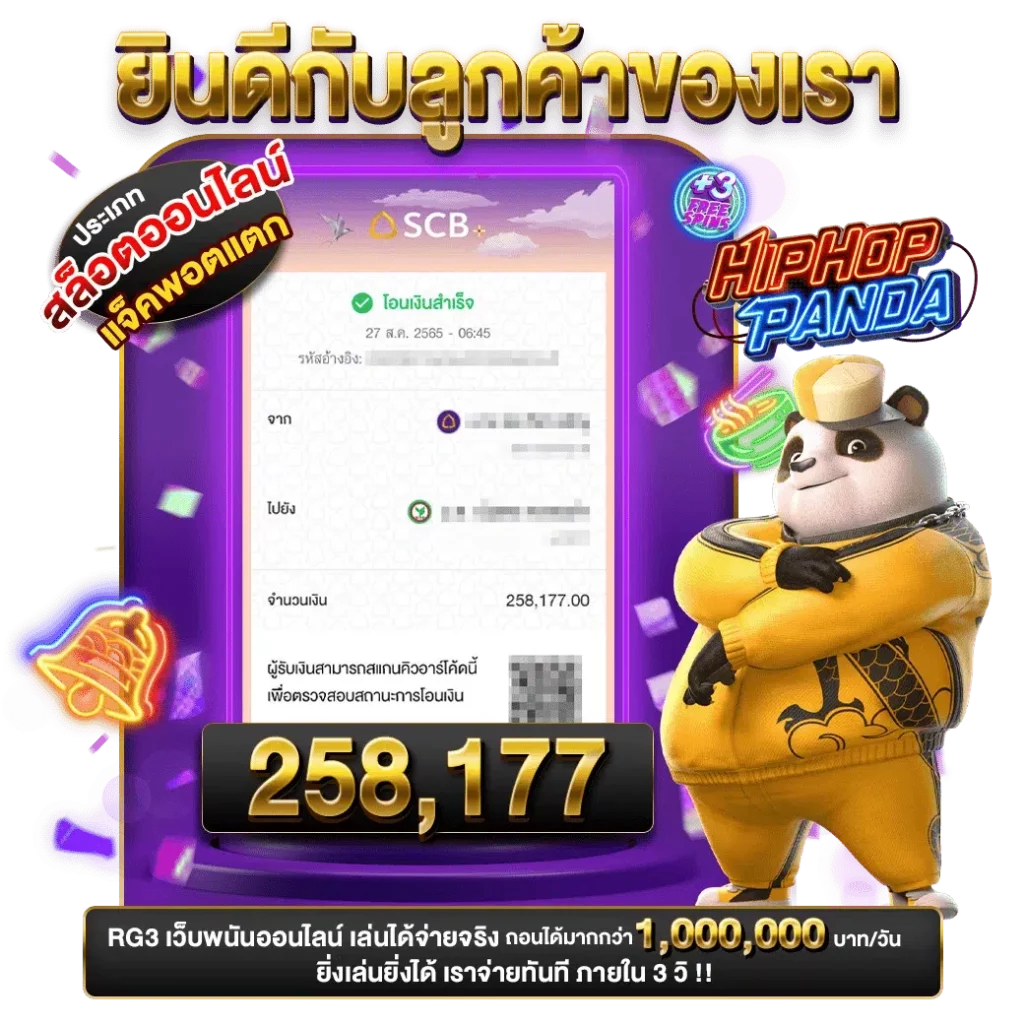
เว็บพนันออนไลน์ที่ปลอดภัยและนิยมในประเทศไทย gb69
gb69 เล่นพนันออนไลน์ที่ปลอดภัยและนิยมในประเทศไทย ทางเรามุ่งมั่นที่จะให้บริการการพนันออนไลน์ที่มั่นใจด้วยการทำธุรกรรมที่รวดเร็วและปลอดภัยทุกขั้นตอน เล่นพนันออนไลน์ที่ทันสมัยและมีคุณภาพกับทีมของเรากับเกมคาสิโนนานาชาติที่ดีที่สุดจากทั่วโลก gb69 slot ทางเรามุ่งมั่นที่จะให้บริการการพนันออนไลน์ที่ปลอดภัย รวดเร็ว และสะดวกสบายในการฝากถอนเพื่อความมั่นใจของผู้เล่นทุกท่านพร้อมนำความสนุกมาสู่คุณโดยตรงตลอด 24 ชั่วโมง
เล่นสนุกถูกใจทั้งวัยรุ่นและคนทำงาน ระบบการพนันออนไลน์ของเรามั่นใจถึงความปลอดภัยในการเงินและการถอนเงินของคุณให้คุณมั่นใจเสมอ บริการการพนันออนไลน์ที่สะดวกสบายและไว้วางใจทำให้เป็นที่นิยมอย่างแพร่หลายในประเทศไทย หากคุณกำลังมองหาเว็บไซต์การพนันที่มั่นคงและสนุก นี่เป็นทางเลือกที่ดีที่สุดสำหรับคุณ อีกทั้งยัง gb69 autobet
เว็บแท้จากอเมริกา ตอบโจทย์เกมพนันออนไลน์

gb69 สล็อต เว็บไซต์เกมพนันออนไลน์ที่เปิดให้เดิมพันสล็อตผ่านมือถือทุกเวลาและรับโบนัส 100% ตลอด 24 ชั่วโมงในปี 2024 สมัครสมาชิกรับโบนัสทันที ไม่มีเงื่อนไข สล็อตออนไลน์ใหม่ 100% ทันที ไม่ต้องคืนแต่ละจำนวน สมาชิกใหม่ที่สมัครสมาชิกเข้าเล่นการพนันออนไลน์จะได้รับโปรโมชั่นพิเศษและโบนัสทุกครั้งที่เข้าเล่น โดยไม่มีเงื่อนไขใดๆ และเข้าเล่นได้ทันทีโดยไม่ต้องรอช้า gb69 เข้าสู่ ระบบ เป็นสถานที่พนันออนไลน์ที่ทันสมัยและเหมาะสำหรับคนที่หลงใหลในการเสี่ยงโชค เว็บแท้จากอเมริกา
เล่นพนันออนไลน์สามารถเปลี่ยนชีวิตของคุณและสร้างความร่ำรวยได้อย่างรวดเร็วในคราวเดียวที่คุณเริ่มต้นเล่นเกมใหม่ การเล่นพนันออนไลน์ในเว็บไซต์ที่มีความปลอดภัยและบริการดีเยี่ยมจะช่วยให้คุณรวยและมั่งคั่งได้อย่างรวดเร็วโดยที่ไม่ต้องกังวลถึงความปลอดภัยและการบริการที่ไม่เบื่อ เว็บพนันที่มีระบบการจ่ายเงินที่ปลอดภัยและเกมสุดน่าสนใจอย่างแท้จริงในประเทศเป็นที่น่าเชื่อถือที่สุดในประเทศ gb69 slot
บทความอื่นๆ ที่น่าสนใจเพิ่มเติม

อุณหภูมิวันนี้
สภาพอากาศวันนี้ กรมอุตุฯ เผย อากาศร้อนถึงร้อนจัด อุณหภูมิสูงสุด 39-41 องศาฯ วันนี้ (ุ6 มี.ค.)
ซานิ
เจออาชีพใหม่ ซานิ AF ที่เจ้าตัวไม่ได้ทำ หลังประวัติส่วนตัวโผล่ วิกิพีเดีย งงเด้! ทำเอานักร้องสาว “ซานิ นิภาภรณ์”

FACE
“อมาโด้ เฟซ” (amado face) รุกตลาดความงาม เปิดตัวผลิตภัณฑ์ใหม่ล่าสุด ผลิตภัณฑ์ “อมาโด้ เฟซ 24เค


